 |
 |
 |
 |
Overview
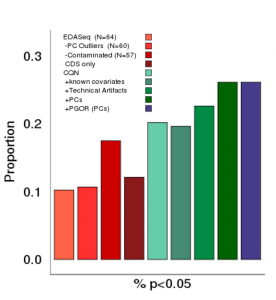
The Arking Lab studies the genomics of complex human disease, with the primary goal of identifying and characterizing genetic variants that modify risk for human disease. Traditional approaches to identify genes involved in complex disease have relied upon screening candidate genes or family-based linkage studies in families with rare monogenic forms of disease. Given the limited success of these approaches to identify genes contributing to common disease, our group has pioneered the use of genome-wide association studies (GWAS), which allow for an unbiased screen of virtually all common genetic variants in the genome. We are currently developing improved GWAS methodology, as well as exploring the integration of additional genome level data (RNA expression, DNA methylation, protein expression) to improve the power to identify genetic influences of disease.
Mitochondrial Copy Number
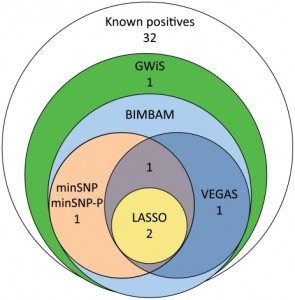
Mitochondrial dysfunction has been well-established as a hallmark of aging and cardiovascular disease. In the Arking lab we have shown mitochondrial DNA copy number (mtDNA CN), a surrogate for mitochondrial function, to be a significant predictor of mortality and incident cardiovascular disease in data from three large prospective studies. We have optimized the calculation mtDNA CN from various types of data, including qPCR, microarray, and next generation sequencing data. Our current research aims to expand upon this finding by identifying genetic determinants of mtDNA CN through a multi-cohort genome-wide association study and characterizing the molecular mechanisms by which mtDNA-CN and heteroplasmy affects cardiovascular disease. Additional research in our lab focuses on exploring the relationship between mtDNA CN with heteroplasmy as well as the link between mtDNA CN and DNA methylation profiles.
Autism
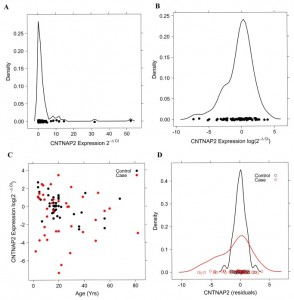
Autism is a devastating neuropsychiatric condition with unknown pathophysiology. Autism spectrum disorders (ASD) have an estimated incidence of 1/200 and thus are more common than many other childhood disorders. Although ASD have a multifactorial etiology, it has a large genetic component, and it is becoming clear that comprehensive efforts involving large sample sizes and methods to reduce heterogeneity are necessary. Our group has recently demonstrated that common sequence changes in the CNTNAP2 and SEMA5A genes are associated with altered risk for ASD. For both these genes, we have been able to demonstrate that gene expression is altered in brains obtained from autism patients relative to age- and sex-matched controls. To identify additional genetic variants, we are performing a comprehensive and integrative screen of autism brains and matched controls, including genetic variation, methylation profiling, and gene expression.
Cardiovascular Genomics
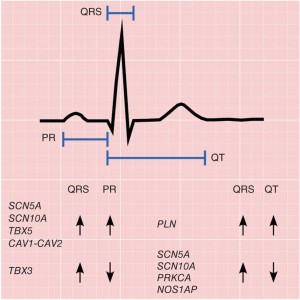
Sudden Cardiac Death (SCD) is one of the leading causes of death in the United States, with ~462,000 of the 2,400,000 (19.3%) US deaths in 1999 classified as SCDs. From the standpoint of preventive care, SCD poses a huge burden since less than 10% of SCD victims survive, and approximately 2/3 of SCD victims do not have clinical symptoms that would warrant preventive intervention. Thus, the use of genetics to identify individuals who are at high risk for SCD is crucial. As a further complication, limited DNA resources are available for individuals who experience SCD due to the low survival rate. We are working to expand the number of available SCD cases, and in addition are collecting hearts from SCD victims for gene expression, DNA methylation, and protein expression profiling. In addition to directly identifying genes for SCD, we are examining intermediate quantitative traits, such as the QT interval, a measure of cardiac repolarization associated with SCD, as well as other ECG parameters. Elucidating the genetic basis of electrical activity in the heart is likely to be a critical component to understanding SCD risk.
For more information about causes, prevention, and research related to SCD risk, please visit the Sudden Cardiac Death Initiative website: pathology.jhu.edu/scd/index.html
Electrophysiology
Electrophysiology is the study of the flow of ions in biological tissues. Our lab is concerned most with electrophysiology of the human heart as assessed by electrocardiography (ECG). Many ECG traits, such as QT interval, are intermediate quantitative traits that predict ventricular arrhythmias and sudden cardiac death (SCD). QT interval in particular could indicate the presence of the potentially lethal Mendelian Long QT Syndrome (LQTS). The QT interval is a measure reflecting myocardial repolarization and has high heritability (Classic twin study: 0.60, 95% CI: 0.49–0.69; PMID: 18031506). Elucidating the genetic basis of electrical activity in the heart is likely to be a critical component to understanding SCD risk.
